TIF 2.0®
EndoGastric Solutions – creating innovative treatment options for GERD
Learn more about TIF® and cTIF™
Treating a broader population of GERD patients
![]()
81% off daily PPIs
![]()
No increased gas or bloating
![]()
30,000+ Procedures
![]()
FDA cleared since 2007
![]()
2797
CE mark since 2006






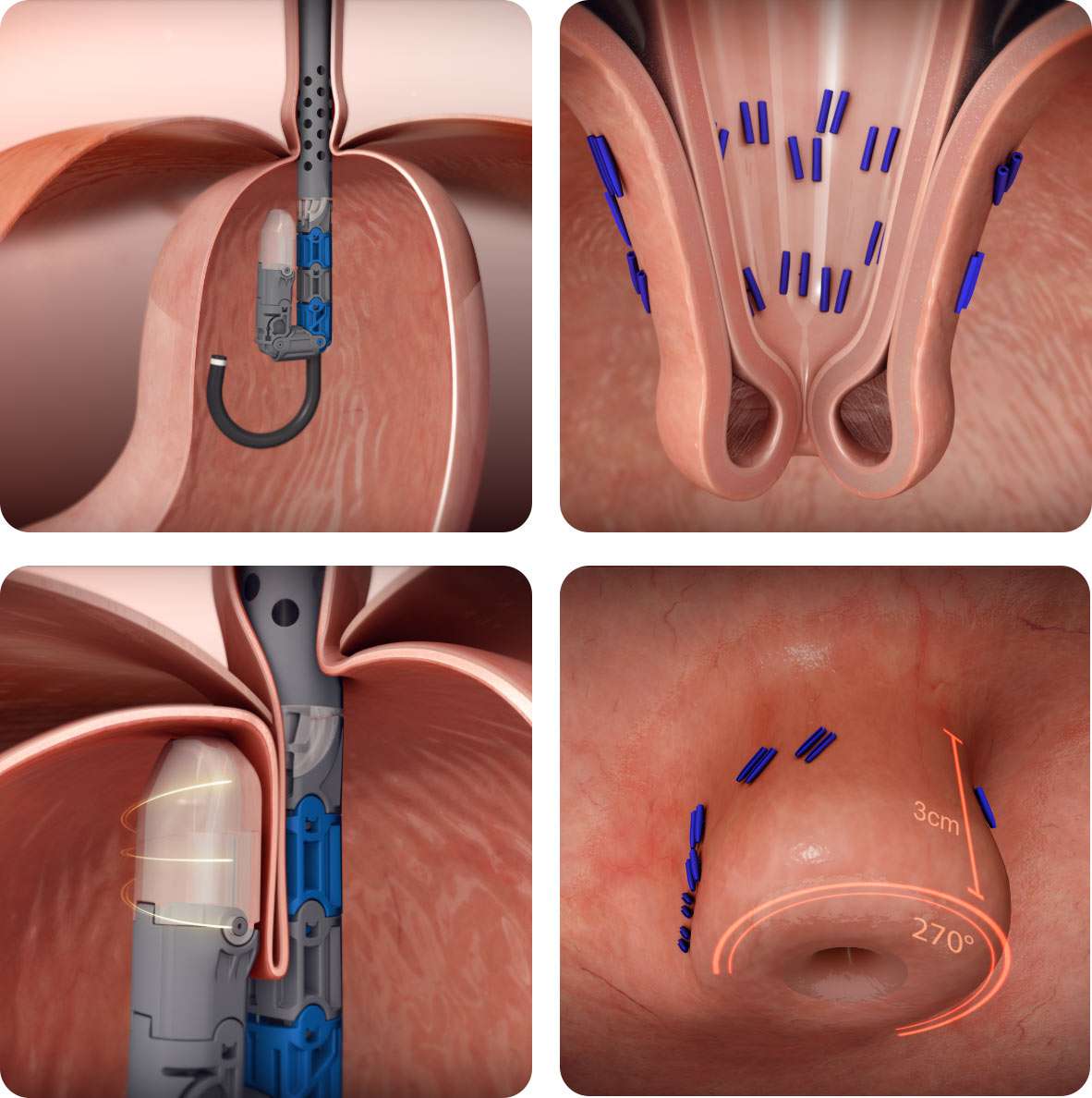
Why TIF 2.0?
- Lower side-effect profile than alternative options
- Restores anatomy to its natural state
- Reproducible and standardizable fundoplication
NP02597-01B